Our scientific studies
Dimpless® efficacy on cellulite and fat metabolism disorders has been demonstrated in two gold-standard clinical trials conducted on healthy women between the ages of 25 and 50 years.
Results showed that through targeted action on subcutaneous fat deposits, Dimpless® is not only able to improve skin appearance and reduce cellulite, but it also accelerates a loss of centimeters.
Results
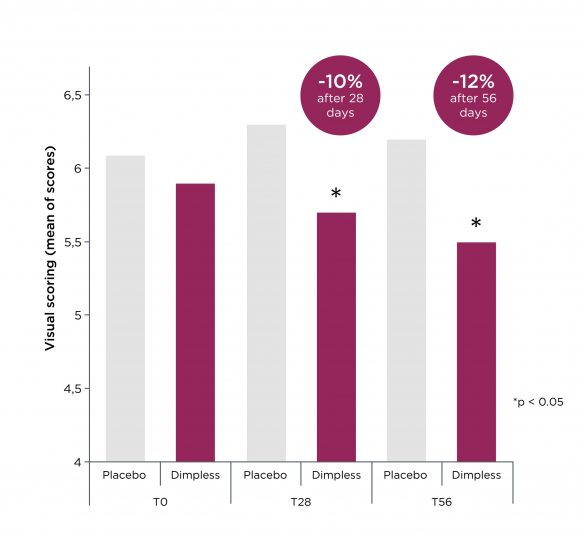
Dimpless® visibly reduces cellulite
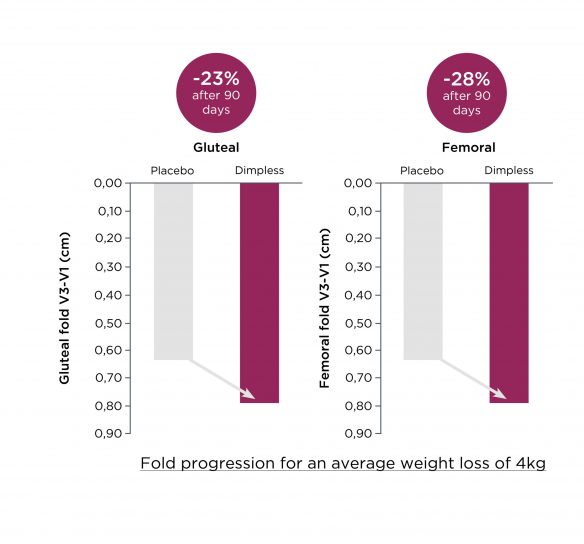
Dimpless® diminue les plis cutanés
Plusieurs mesures de plis cutanés ont également été réalisées, dans des zones spécifiquement concernées par le stockage des graisses et la cellulite, à savoir les fesses (pli fessier) et les cuisses (pli fémoral).
Les résultats ont mis en évidence une diminution du pli fessier et fémoral versus Placebo, avec respectivement -23% et -28% après 3 mois de supplémentation.

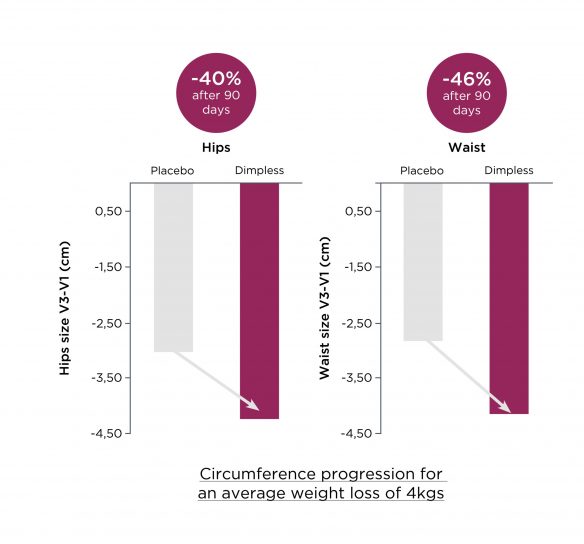
Dimpless® accélère la diminution du tour de taille et de hanches
Dimpless® efficacy on subcutaneous fat deposits and visible cellulite relies on its
multi-parametric effect on fat metabolism.
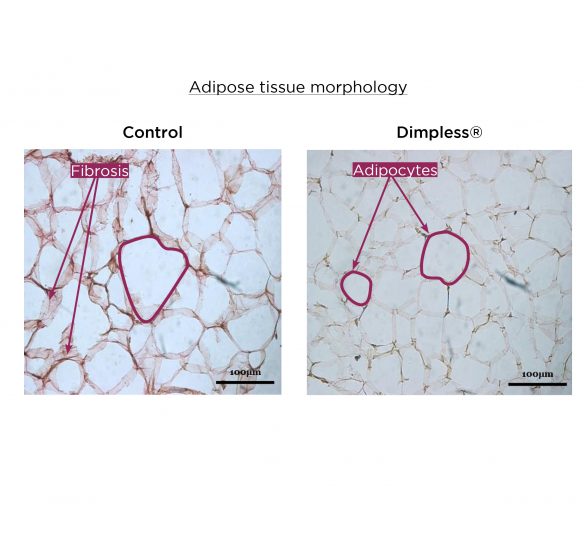
Adipocytes size reduction and collagen fibrosis resorption
While metabolic disorders may induce adipocyte hypertrophy and fibrosis of collagen fibers, Dimpless® has been shown to significantly improve adipose tissue morphology.
After supplementation, both the, cell area and fibrosis decreased by over 50% compared to control.
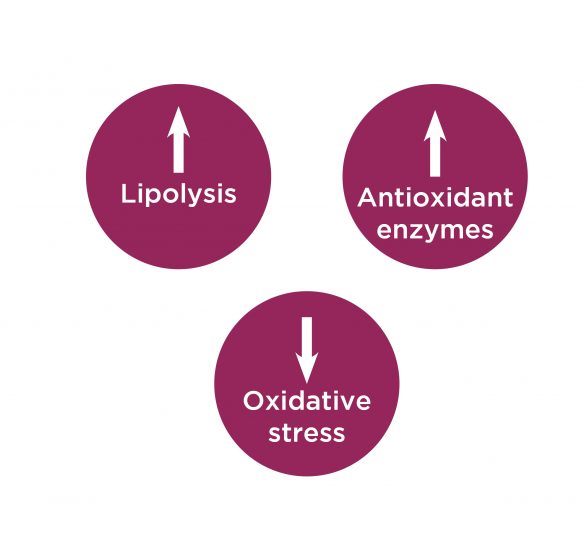
Antioxidant capacity and lipolysis activation
Fat storage is known to alter the oxidative state.
The expansion of adipose tissue is associated with both an induction of free radicals through the activation of NADPH oxidase and a decrease in endogenous antioxidant enzymes.
Dimpless® supplementation has been shown to normalize the expression of SOD, Catalase, and Glutathione Peroxidase, and also restore lipolysis to a healthy level.
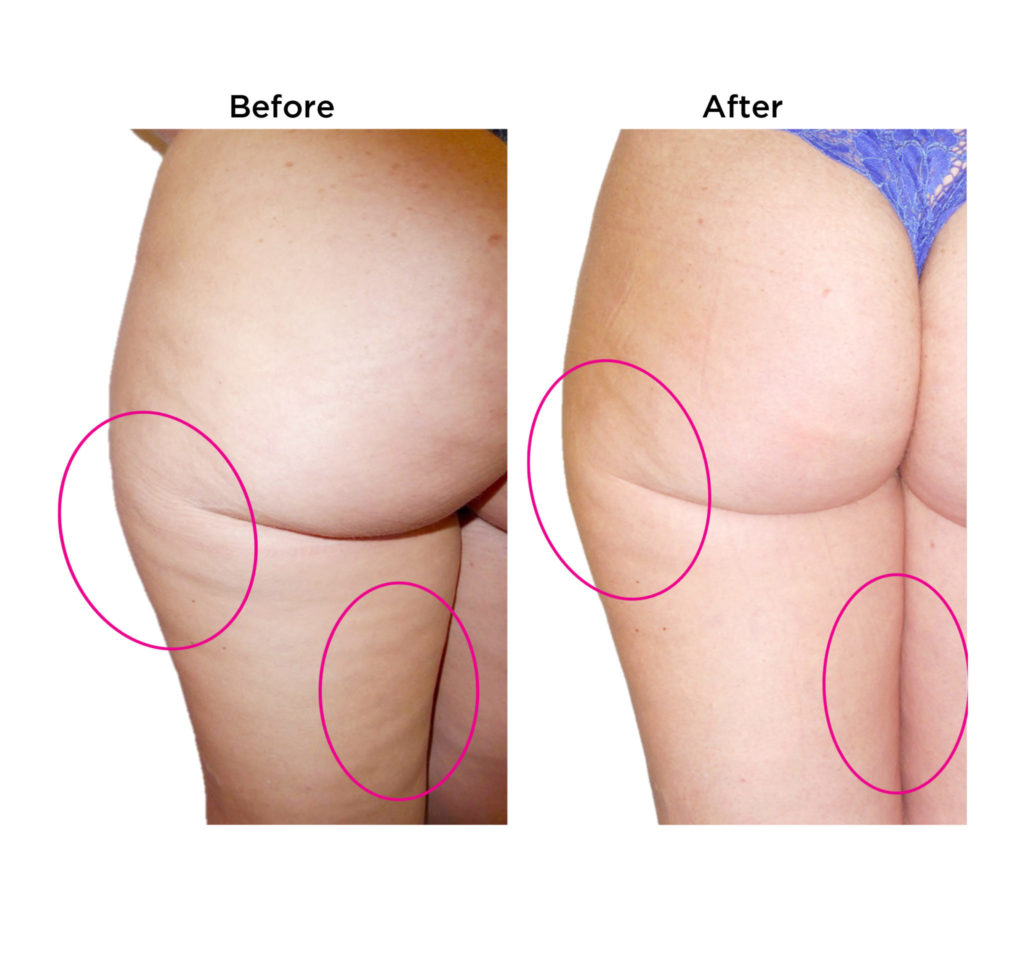
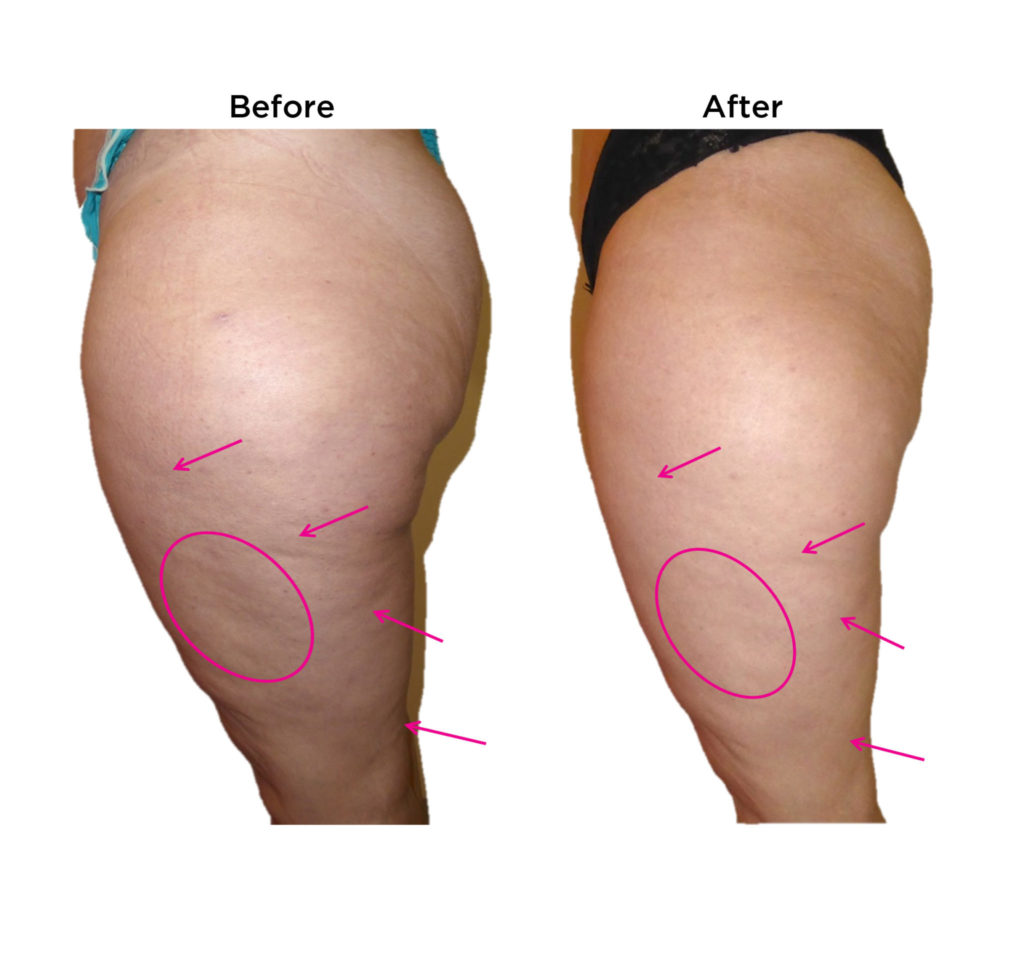
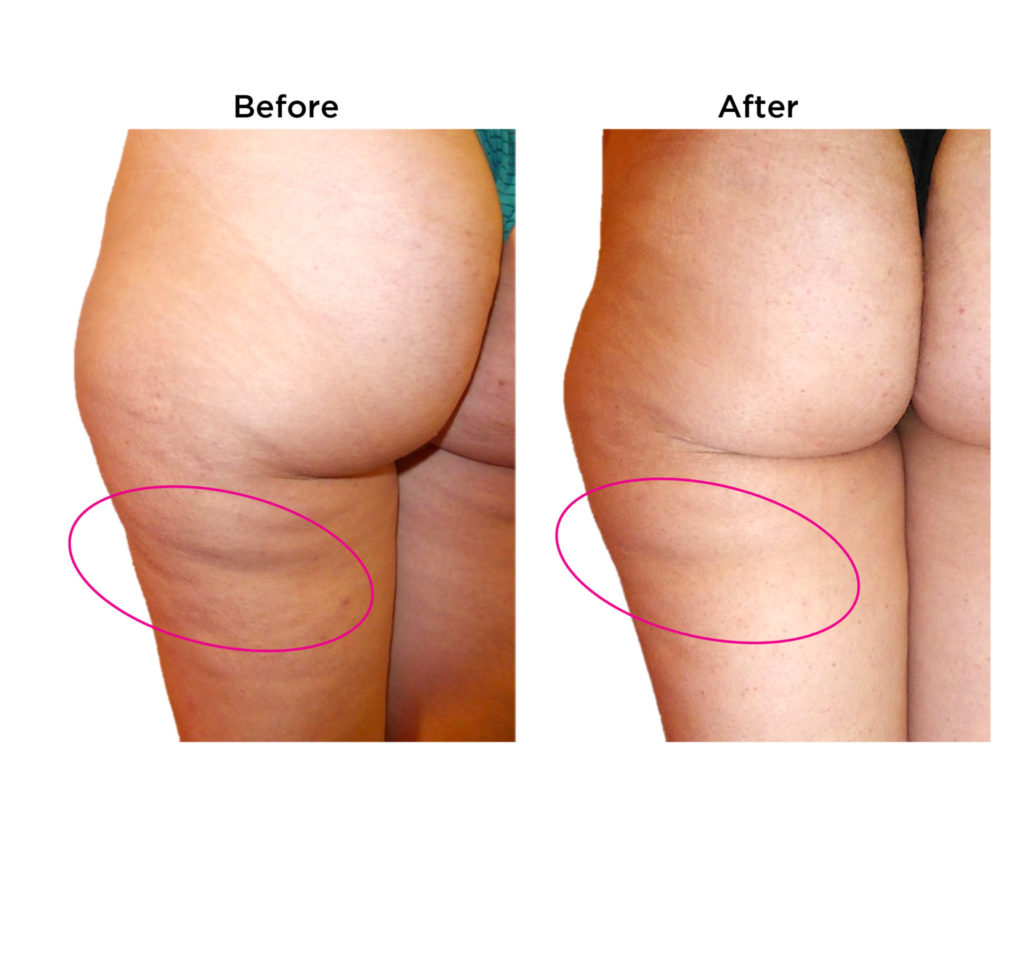
These results were observed in
gold-standard clinical trials.
Lemaire & al, Phytothérapie 2015
Clinical trial of a natural and bioactive melon SuperOxide Dismutase
(SOD B Dimpless®) on cellulite.
Randomized, double-blind, placebo-controlled
41 women/31 – 50 years old/
40mg (480 UI SOD)/2 months
Unpublished, 2019
Randomized, double-blind, placebo-controlled
35 overweight women (25<IMC<31)/25-50 ans/40mg (480 UI SOD)/3 months